 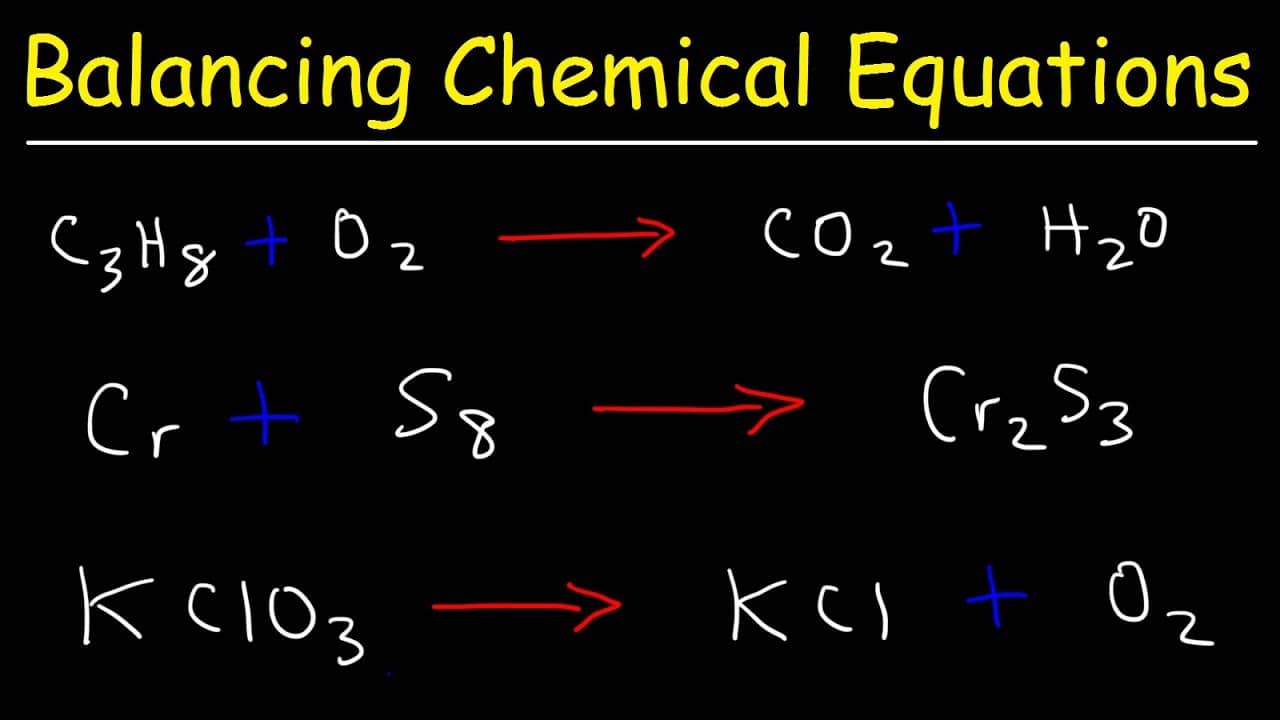
Look no further to know how to find the theoretical yield:įirst, calculate the moles of your limiting reagent. The measurements you need are the mass of the reagents, their molecular weights, the stoichiometry of the reaction (found from the balanced equation), and the molecular weight of the desired product. Now, the theoretical yield formula may seem challenging to understand, so we will show you a quick guide on how to calculate the theoretical yield. Remember to hit refresh at the bottom of the calculator to reset it. If you are uncertain which of your reagents are limiting, plug in your reagents one at a time, and whichever one gives you the lowest number of moles is the limiting reagent. IMPORTANT NOTE: Yields can only be found using the limiting reagent. You can also use the theoretical yield equation to ensure that you react with equal moles of your reactants so no molecule is wasted. This allows you to work out how efficiently you carried out your reaction (the quantity you can find at the actual yield calculator), which is done by calculating the percent yield. So, Oxygen atoms are not balanced at the products side.This theoretical yield calculator will answer all the burning questions you have regarding how to calculate the theoretical yield, such as how to find theoretical yield as well as the theoretical yield definition and the theoretical yield formula.īefore carrying out any kind of lab work, you need to work out what is the theoretical yield so you know how much of your product, be it a molecule or lattice, you can expect from a given amount of starting material. A balanced equation is nothing but the equation where number of atoms in each element in reactants and products are same. What does meant by balancing chemical equations?Ĭhemical Equation is defined as the symbolic representation of chemical reaction. Replace those variable values in place of coefficients in the actual equation to get it balanced.Ĥ.Substitute the random values in the equation by using trial and error method, we can find the variable values.Equate any one element number of molecules on both sides.Take a note of number of elements on the right hand side and left hand side.At first, add variables a,b,c,d, so on as coefficients to all the reactants and products.How to balance long chemical equation using algebraic method? Always multiply the coefficients of the atoms by its subscript to identify the number of elements in the reaction.ģ. The important point to remember while balancing equation is never change its subscripts, add coefficients to them. Do you add or multiply when balancing equations? You are not supposed to add subscripts, because it will change the reaction.Ģ. Balance the chemical equation by putting coefficients in front of them.Once balancing of the one element is completed, proceed further until all elements are balanced in the equation.Start by balancing an element that is available in only one reactant and product.Apply the Law of Conservation of Mass to get the same number of atoms on each side of the chemical equation.What are the rules for balancing the chemical equation? Have a look at them and use whenever required to get the instant results.ġ. is a best place where you can find the calculators of various math, physics, and chemistry concepts. Nitrogen, Chlorine and Oxygen atoms are not balanced. Right hand side has Ca= 1, Cl= 1, Ag= 1, N= 2, O= 6 Left hand side has Ca=1, Cl= 2, Ag= 1, N= 1, O= 3 Write down the number of atoms in each element Question: Balance this equation: CaCl 2 + AgNO 3 → Ca(NO 3) 2 + AgCl? Sometimes, count all the atoms in the chemical equation even if it has poly atomic ions and add coefficients to it to get the balanced equation.Add coefficients to those items to get the balanced equation.When you are dealing with poly atomic ions, treat every poly atomic ion as one item.Change the coefficients for the required atoms to get the equation balanced.After adding the coefficients once check the number of atoms on left side are equal to number of atoms on right side.You are just allowed to place coefficient in front of the reactant or product, but not allowed to change subscripts of the elements.Check the coefficients of atoms and add any number n as coefficient for the required atoms to balance the equation.Count the number of atoms on each side of the equation.Follow these guidelines and balance your equation easily and quickly. Here we are giving the 5 simple steps to balance any type of chemical equation.
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
